June 18, 2016
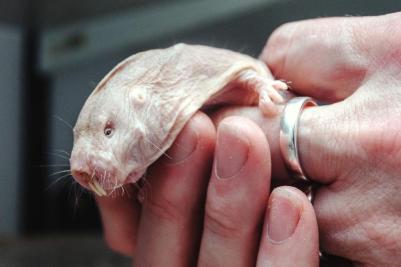
Dr. Chris Faulkes is standing in his laboratory, tenderly caressing what looks like a penis. It’s not his penis, nor mine, and it’s definitely not that of the only other man in the room, VICE photographer Chris Bethell. But at four inches long with shrivelled skin that’s veiny and loose, it looks very penis-y. Then, with a sudden squeak, it squirms in his hand as if trying to break free, revealing an enormous set of Bugs Bunny teeth protruding from the tip.
“This,” says Faulkes, “is a naked mole rat, though she does look like a penis with teeth, doesn’t she? Or a saber-tooth sausage. But don’t let her looks fool you—the naked mole rat is the superhero of the animal kingdom.”
I’m with Faulkes in his lab at Queen Mary, University of London. Faulkes is an affable guy with a ponytail, telltale tattoos half-hidden under his T-shirt sleeve, and a couple of silver goth rings on his fingers. A spaghetti-mess of tubes weave about the room, like a giant gerbil maze, through which 12 separated colonies of 200 naked mole rats scurry, scratch, and squeak. What he just said is not hyperbole. In fact, the naked mole rat shares more than just its looks with a penis: Where you might say the penis is nature’s key to creating life, this ugly phallus of a creature could be mankind’s key to eternal life.
“Their extreme and bizarre lifestyle never ceases to amaze and baffle biologists, making them one of the most intriguing animals to study,” says Faulkes, who has devoted the past 30 years of his life to trying to understand how the naked mole rat has evolved into one of the most well-adapted, finely tuned creatures on Earth. “All aspects of their biology seem to inform us about other animals, including humans, particularly when it comes to healthy aging and cancer resistance.”
Similarly sized rodents usually live for about five years. The naked mole rat lives for 30. Even into their late 20s, they hardly seem to age, remaining fit and healthy with robust heartbeats, strong bones, sharp minds, and high fertility. They don’t seem to feel pain, and, unlike other mammals, they almost never get cancer.
In other words, if humans lived as long, relative to body size, as naked mole rats, we would last for 500 years in a 25-year-old’s body. “It’s not a ridiculous exaggeration to suggest we can one day manipulate our own biochemical and metabolic pathways with drugs or gene therapies to emulate those that keep the naked mole rat alive and healthy for so long,” says Faulkes, stroking his animal. “In fact, the naked mole rat provides us the perfect model for human aging research across the board, from the way it resists cancer to the way its social systems prolong its life.”
Over the centuries, a long line of optimists, alchemists, hawkers, and pop stars have hunted various methods of postponing death, from drinking elixirs of youth to sleeping in hyperbaric chambers. The one thing those people have in common is that all of them are dead. Still, the anti-aging industry is bigger than ever. In 2013, its global market generated more than $216 billion. By 2018, it will hit $311 billion, thanks mostly to huge investment from Silicon Valley billionaires and Russian oligarchs who’ve realized the only way they could possibly spend all their money is by living forever. Even Google wants in on the action, with Calico, its $1.5 billion life-extension research center whose brief is to reverse-engineer the biology that makes us old or, as Time magazine put it, to “cure death.” It’s a snowballing market that some are branding “the internet of healthcare.” But on whom are these savvy entrepreneurs placing their bets? After all, the race for immortality has a wide field.
In an office not far from Google’s headquarters in Mountain View, with a beard to his belt buckle and a ponytail to match, British biomedical gerontologist Aubrey De Grey is enjoying the growing clamor about conquering aging, or “senescence,” as he calls it. His charity, the SENS Research Foundation, has enjoyed a bumper few years thanks to a $600,000-a-year investment from Paypal co-founder and immortality motormouth Peter Thiel (“Probably the most extreme form of inequality is between people who are alive and people who are dead”). Though he says the foundation’s $5.75 million annual budget can still “struggle” to support its growing workload.
According to the Cambridge-educated scientist, the fundamental knowledge needed to develop effective anti-aging therapies already exists. He argues that the seven biochemical processes that cause the damage that accumulates during old age have been discovered, and if we can counter them we can, in theory, halt the aging process. Indeed, he not only sees aging as a medical condition that can be cured, but believes that the “first person to live to 1,000 is alive today.” If that sounds like the ramblings of a crackpot weird-beard, hear him out; Dr. De Grey’s run the numbers.
“If you look at the math, it is very straightforward,” he says. “All we are saying here is that it’s quite likely that within the next twenty or thirty years, we will develop medicines that can rejuvenate people faster than time is passing. It’s not perfect yet, but soon we’ll take someone aged sixty and fix them up well enough that they won’t be sixty again, biologically, for another thirty years. In that period, therapies will improve such that we’ll be able to rejuvenate them again, so they won’t be sixty for a third time until they are chronologically one hundred fifty, and so on. If we can stay one step ahead of the problem, people won’t die of aging anymore.”
“Like immortality?” I ask. Dr. De Grey sighs: “That word is the bane of my life. People who use that word are essentially making fun of what we do, as if to maintain an emotional distance from it so as not to get their hopes up. I don’t work on ‘curing death.’ I work on keeping people healthy. And, yes, I understand that success in my work could translate into an important side effect of people living longer. But to ‘cure death’ implies the elimination of all causes, including, say, dying in car accidents. And I don’t think there’s much we could do to survive an asteroid apocalypse.”
So instead, De Grey focuses on the things we can avoid dying from, like hypertension, cancer, Alzeimer’s, and other age-related illnesses. His goal is not immortality but “radical life extension.” He says traditional medicines won’t wind back the hands of our body clocks—we need to manipulate our makeup on a cellular level, like using bacterial enzymes to flush out molecular “garbage” that accumulates in the body, or tinkering with our genetic coding to prevent the growth of cancers, or any other disease.
Chris Faulkes knows of one magic bullet to kill cancer. And, back at Queens, he is making his point by pulling at the skin of a naked mole rat in his hand. “It’s the naked mole rat’s elasticky skin that’s made it cancer-proof,” he says. “The theory—first discovered by a lab in America—is that, as an adaptation to living underground in tight tunnels, they’ve developed a really loose skin so they don’t get stuck or snagged. That elasticity is a result of it producing this gloopy sugar [polysacharide], high-molecular-weight hyaluronan (HMW-HA).”
While humans already have a version of hyaluronan in our bodies that helps heal wounds by encouraging cell division (and, ironically, assist tumor growth), that of the naked mole rat does the opposite. “The hyaluronan in naked mole rats is about six times larger than ours,” says Faulkes. “It interacts with a metabolic pathway, which helps prevent cells from coming together to make tumors.”
But that’s not all: It is believed it may also act to help keep their blood vessels elastic, which, in turn, relieves high blood pressure (hypertension)—a condition that affects one in three people and is known in medical circles as “the silent killer” because most patients don’t even know they have it. “I see no reason why we can’t use this to inform human anti-cancer and aging therapies by manipulating our own hyaluronan system,” says Faulkes.
Then there are the naked mole rat’s cells themselves, which seem to make proteins – the molecular machines that make bodies work—more accurately than ours, preventing age-related illnesses like Alzheimer’s. And the way they handle glucose doesn’t change with age either, reducing their susceptibility to things like diabetes. “Most of the age-related declines you see in the physiology in mammals do not occur in naked mole rats,” adds Faulkes. “We’ve only just begun on the naked mole rat story, and already a whole universe is opening up that could have a major downstream effect on human health. It’s very exciting.”
Of course, the naked mole rat isn’t the only animal scientists are probing to pick the lock of long life. “With a heart rate of 1,000 beats a minute, the tiny hummingbird should be riddled with rogue free radicals [the oxygen-based chemicals that basically make mammals old by gradually destroying DNA, proteins and fat molecules]… but it’s not,” says zoologist Jules Howard, author of Death on Earth: Adventures in Evolution and Mortality. “Then there are pearl mussel larvae that live in the gills of Atlantic salmon and mop up free radicals, and lobsters, which seem to have evolved a protein which repairs the tips of DNA [telomeres], allowing for more cell divisions than most animals are capable of. And we mustn’t forget the 2mm-long C. elegans roundworm. Within these 2mm-long nematodes are genetic mechanisms that can be picked apart like cogs and springs in an attempt to better understand the causes of aging and ultimately death.”
But there is one animal on Earth that may hold the master key to immortality: the Turritopsis dohrnii, or Immortal Jellyfish. Most jellyfish, when they reach the end of life, die and melt into the sea. Not the Turritopsis dohrnii. Instead, the 4mm sea creature sinks to the bottom of the ocean floor, where its body folds in on itself—assuming the jellyfish equivalent of the fetal position—and regenerates back into a baby jellyfish, or polyp, in a rare biological process called transdifferentiation, in which its old cells essentially transform into young cells.
There is just one scientist who has been culturing Turritopsis polyps in his lab consistently. He works alone, without major financing or a staff, in a poky office in Shirahama, a sleepy beach town near Kyoto. Yet professor Shin Kubota has managed to rejuvenate one of his charges 14 times, before a typhoon washed it away. “The Turritopsis dohrnii is a miracle of nature,” he says over the phone. “My ultimate purpose is to understand exactly how they regenerate so we can apply its mechanisms to human beings. You see, very surprisingly, the Turritopsis’s genome is very similar to humans’—much more so than worms. I believe we will have the technology to begin applying this immortal genome to humans very soon.”
How soon? “In 20 years,” he says, a little mischievously. “That is my guess.”
If PKubota really believes his own claim, then he’s got a race on his hands; he’s not the only scientist with a “20-year” prophesy. The acclaimed futurist and computer scientist Ray Kurzweil believes that by the 2030s we’ll have microscopic machines traveling through our bodies, repairing damaged cells and organs, effectively wiping out diseases and making us biologically immortal anyway. “The full realization of nanobots will basically eliminate biological disease and aging,” he told the world a few years back.
It’s a blossoming industry. And, in a state-of-the-art lab at the Bristol Robotics Laboratory, at Bristol University, Dr. Sabine Hauert is on its coalface. She designs swarms of nanobots—each a thousand times smaller than the width of a hair—that can be injected into the bloodstream with a payload of drugs to infiltrate the pores of cancer cells, like millions of tiny Trojan Horses, and destroy them from within. “We can engineer nanoparticles to basically do what we want them to do,” she tells me. “We can change their size, shape, charge, or material and load them with molecules or drugs that they can release in a controlled fashion.”
While she says the technology can be used to combat a whole gamut of different illnesses, Dr. Hauert has trained her crosshairs on cancer. What’s the most effective nano-weapon against malignant tumors? Gold. Millions of swarming golden nanobots that can be dispatched into the bloodstream, where they will seep into the tumor through little holes in its rapidly-growing vessels and lie in wait. “Then,” she says, “if you heat them with an infrared laser they vibrate violently, degrading the tumour’s cells. We can then send in another swarm of nanoparticles decorated with a molecule that’s loaded with a chemotherapy drug, giving a 40-fold increase in the amount of drugs we can deliver. This is very exciting technology that is already having a huge impact on the way we treat cancer, and will do on other diseases in the future.”
The next logical step, as Kurzweil claims, is that we will soon have nanobots permanently circulating in our veins, cleaning and maintaining our bodies indefinitely. They may even replace our organs when they fail. Clinical trials of such technology is already beginning on mice.
The naked mole rat colony in Chris Faulkes’s lab
The oldest mouse ever to live was called Yoda. He lived to the age of four. The oldest ever dog, Bluey, was 29. The oldest flamingo was 83. The oldest human was 122. The oldest clam was 507. The point is, evolution has rewarded species who’ve worked out ways to not get eaten by bigger species—be it learning to fly, developing big brains or forming protective shells. Naked mole rats went underground and learned to work together.
“A mouse is never going to worry about cancer as much as it will about cats,” says Faulkes. “Naked mole rats have no such concerns because they built vast networks of tunnels, developed hierarchies and took up different social roles to streamline productivity. They bought themselves time to evolve into biological marvels.”
At the top of every colony is a queen. Second in rank are her chosen harem of catamites with whom she mates for life. Beneath them are the soldiers and defenders of the realm, the biggest animals around, and at the bottom are the workers who dig tunnels with their teeth or search for tubers, their main food source. They have a toilet chamber, a sleeping chamber, a nursing chamber and a chamber for disposing of the dead. They rarely go above ground and almost never mix with other colonies. “It’s a whole mosaic of different characteristics that have come about through adapting to living in this very extreme ecological niche,” says Faulkes. “All of the weird and wonderful things that contribute to their healthy aging have come about through that. Even their extreme xenophobia helps prevent them being wiped out by infectious diseases.”
Still, the naked mole rat is not perfect. Dr. Faulkes learned this the hard way one morning in March last year, when he turned the light on in his lab to a grisly scene. “Blood was smeared about the perspex walls of a tunnel in colony N,” he says, “and the mangled corpse of one of my mole rats lay lifeless inside.” There was one explanation: A queen had been murdered. “There had been a coup,” he recalls. “Her daughter had decided she wanted to run the colony so she savaged her mother to death to take over. You see, naked mole rats may be immune to death by aging, but they can still be killed, just like you and me.”
That’s the one issue that true immortalists have with the concept of radical life extension: we can still get hit by a bus or murdered. But what if the entire contents of your brain—your memories, beliefs, hopes, and dreams—could be scanned and uploaded onto a mainframe, so when You 1.0 finally does fall down a lift shaft or is killed by a friend, You 2.0 could be fed into a humanoid avatar and rolled out of an immortality factory to pick up where you left off?
Dr. Randall Koene insists You 2.0 would still be you. “What if I were to add an artificial neuron next to every real neuron in your brain and connect it with the same connections that your normal neurons have so that it operates in exactly the same way?” he says. “Then, once I’ve put all these neurons in place, I remove the connections to all the old neurons, one by one, would you disappear?”
More at: https://www.vice.com/read/quest-for-immortality-what-will-win-tech-animals